Need help? We're here to assist you!
Thank You for Enquiry, we will contact you soon!
Close
The Class 8 is an important year in a student’s life and Science is one of the subjects that require dedication, hard work, and practice. It’s a subject where you can score well if you are well-versed with the concepts, remember the important formulas and solving methods, and have done an ample amount of practice. Worry not! Home Revise is here to make your Class 8 journey even easier. It’s essential for students to have the right study material and notes to prepare for their board examinations, and through Home Revise, you can cover all the fundamental topics in the subject and the complete NCERT Class 8 Science Book syllabus.

Exercise Questions
1. Which of the following can be beaten into thin sheets?
(a) Zinc
(b) Phosphorus
(c) Sulphur
(d) Oxygen
Solution:
Answer is a) Zinc
Explanation:
Here, zinc is a metal with malleability and ductility whereas Phosphorus, Sulphur and Oxygen are nonmetals which lack malleability and ductility.
2. Which of the following statements is correct?
(a) All metals are ductile.
(b) All non-metals are ductile.
(c) Generally, metals are ductile.
(d) Some non-metals are ductile.
Solution:
Answer is (c) Generally, metals are ductile.
Explanation:
Ductility is a property where a substance can be drawn into thin wires. Generally, metals are ductile with mercury as the exception.
3. Fill in the blanks.
(a) Phosphorus is a very _________non-metal.
(b) Metals are _________ conductors of heat and ____________ .
(c) Iron is ____________reactive than copper.
(d) Metals react with acids to produce ____________ gas.
Solution:
(a) Phosphorus is a very reactive non-metal.
(b) Metals are good conductors of heat and electricity.
(c) Iron is more reactive than copper.
(d) Metals react with acids to produce hydrogen gas.
4. Mark ‘T’ if the statement is true and ‘F’ if it is false.
(a) Generally, non-metals react with acids. ( )
(b) Sodium is a very reactive metal. ( )
(c) Copper displaces zinc from zinc sulphate solution. ( )
(d) Coal can be drawn into wires. ( )
Solution:
a) False
b) True
c) False
d) False
5. Some properties are listed in the following table. Distinguish between metals and non-metals on the basis of these properties.
| Properties | Metals | Non-metals |
| 1. Appearance | ||
| 2. Hardness | ||
| 3. Malleability | ||
| 4. Ductility | ||
| 5. Heat Conduction | ||
| 6. Conduction of Electricity |
Solution:
| Properties | Metals | Non-metals |
| 1. Appearance | Lustrous | Dull |
| 2. Hardness | Hard | Soft |
| 3. Malleability | Have property of Malleability | Do not have a property of Malleability |
| 4. Ductility | Have property of Ductility | Do not have the property of Ductility |
| 5. Heat Conduction | Good conductor of Heat | Bad Conductor of Heat |
| 6. Conduction of Electricity | Good conductor of Electricity | The bad conductor of Electricity |
6. Give reasons for the following.
(a) Aluminium foils are used to wrap food items.
(b) Immersion rods for heating liquids are made up of metallic substances.
(c) Copper cannot displace zinc from its salt solution.
(d) Sodium and potassium are stored in kerosene
Solution:
a) Aluminium is malleable and can be drawn into thin sheets; hence aluminium foils are used to wrap food items
b) Immersion rods for heating liquids are made up of metallic substances because metals are good conductors of heat and electricity.
c) Copper cannot displace zinc from its salt solution because zinc is more reactive than copper.
d) Sodium and Potassium are highly reactive metals which readily reacts with atmospheric oxygen to catch fire; hence sodium and potassium are stored in kerosene.
7. Can you store lemon pickle in an aluminium utensil? Explain.
Solution:
Pickle contains acids which react with aluminium metal to produce salt and hydrogen. Hence pickle is not stored in aluminium utensil.
8. Match the substances given in Column A with their uses given in Column B.
| A | B |
| (i) Gold | (a) Thermometers |
| (ii) Iron | (b) Electric wire |
| (iii) Aluminium | (c) Wrapping food |
| (iv) Carbon | (d) Jewellery |
| (v) Copper | (e) Machinery |
| (vi) Mercury | (f) Fuel |
Solution:
| A | B |
| (i) Gold | (d) Jewellery |
| (ii) Iron | (e) Machinery |
| (iii) Aluminium | (c) Wrapping food |
| (iv) Carbon | (f) Fuel |
| (v) Copper | (b) Electric wire |
| (vi) Mercury | (a) Thermometers |
9. What happens when
(a) Dilute sulphuric acid is poured on a copper plate?
(b) Iron nails are placed in a copper sulphate solution?
Write word equations of the reactions involved.
Solution:
(i) No reaction occurs when dilute sulphuric acid is poured on a copper plate. However, when concentrated sulphuric acid is poured on a copper plate, hydrogen gas evolves along with the formation of blue coloured copper sulphate crystals. The chemical reaction for the reaction between concentrated sulfuric acid and copper is:
Cu + H2
SO4
(conc.) ->CuSO4
+ H2
ii) Iron, being more reactive, displaces copper from copper sulphate. In this reaction, the blue colour of copper sulphate fades and there is a deposition of copper on the iron nail.
Fe + CuSO4 → FeSO4 + Cu
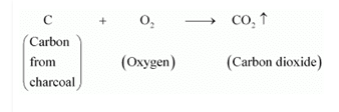
10. Saloni took a piece of burning charcoal and collected the gas evolved in a test tube.
(a) How will she find the nature of the gas?
(b) Write down word equations of all the reactions taking place in this process.
Solution:
a) In a test tube containing gas, add a few drops of water. Now cover the test tube and shake well. After shaking, test the solution with blue litmus. It will change from blue to red. Thus, gas is acidic in nature.
b) Charcoal reacts with oxygen to form carbon dioxide gas.
11. One day Reeta went to a jeweller’s shop with her mother. Her mother gave old gold jewellery to the goldsmith to polish. Next day when they brought the jewellery back, they found that there was a slight loss in its weight. Can you suggest a reason for the loss in weight?
Solution:
In order to polish the gold ornament, it is to be dipped into a liquid called aqua regia (a mixture of hydrochloric acid and nitric acid). On getting immersed in aqua regia, the outer layer of gold dissolves and an inner shiny layer appears. The dissolving of the layer causes a reduction in the weight of the jewellery.